|
That means some of sulfide’sĮlectrons are more distant from the nucleus, so sulfide has aĮ. Both are in the same group, but sulfide anion has moreĮnergy levels with filled orbitals. That means oxide’s outermostĮlectrons will be farther from its nucleus, so oxide has a smallerĭ. Both are in the same period, but oxide anion has fewerĮnergy levels with filled orbitals. That means some of oxide’sĮlectrons are more distant from the nucleus, so oxide has a smallerĬ. Both are in the same group, but oxide anion has moreĮnergy levels with filled orbitals. Towards the nucleus, so sulfide has a smaller radius.ī. 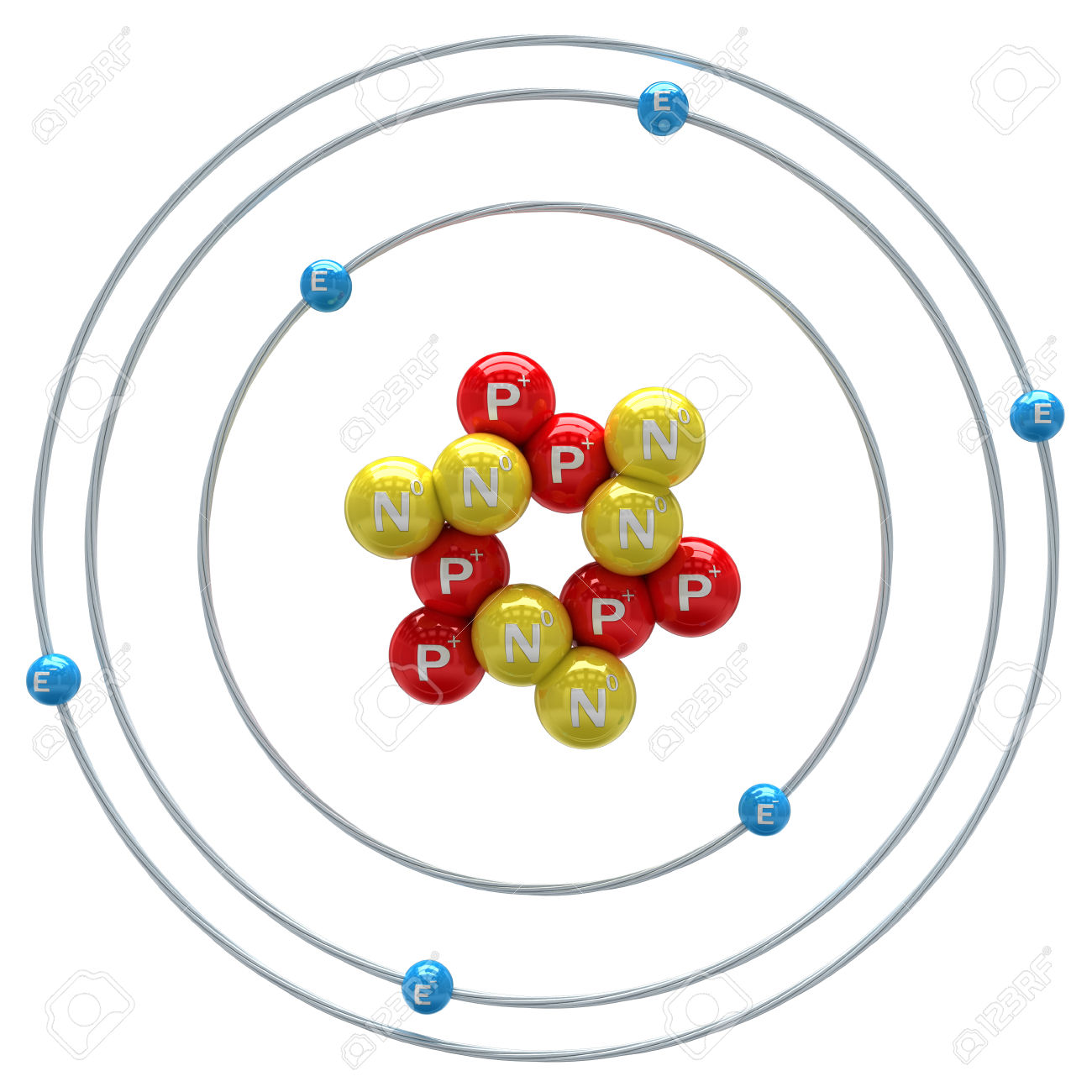
That means its electrons are pulled with greater strength Both are in the same group, but sulfide anion has more Identifies their size difference and justifies its fundamentalĪ. The ionic radius of the oxygen 2- anion is much different than Both are in the same period, but oxygen has a higherġ year ago The atomic radius of oxygen is smaller than that of carbon. Which results in an atom of smaller size.Į. With more strength, hence the electronic cloud is more compressed, Both are in the same period, but oxygen is moreĮlectronegative than carbon. More strongly attracted to its nucleus, hence the electronic cloudĭ. Both are in the same period, but oxygen has a lower Is more compressed, which results in a smaller size.Ĭ. Both are in the same period, but oxygen has a higher 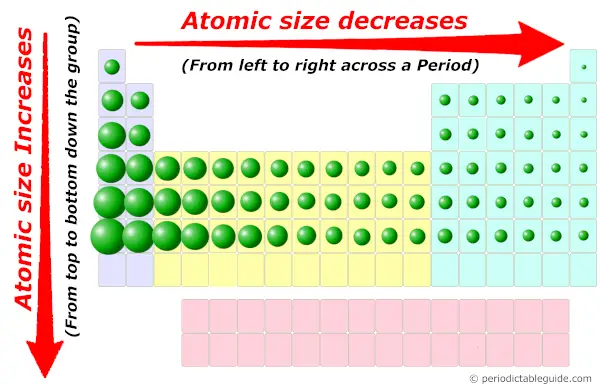
Is less compressed, which results in a smaller size.ī. More loosely attracted to its nucleus, hence the electronic cloud SOLVED: The atomic radius of oxygen is smaller than that of carbon.Ĭhoose the one answer that properly justifies the fundamental causeĪ.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
